EXTEDO eCTDmanager
eCTDmanager – eCTD Managerment Electronic Submission Solution
As the trusted validator used by more than 35 regulatory authorities, eCTDmanager goes beyond basic eCTD submissions software. It also provides you with a complete regulatory dossier assembly environment that enables your organization to operate in a compliant manner within a heavily regulated environment. With eCTDmanager, you are able to build, view, validate and publish compliant submissions based on eCTD, NeeS, eCopy, IMPD, CTA, MAA、VNeeS, DMF, ASMF and other regional formats.
The effective management of validated and compliant eSubmissions is a complex process. The need to build, view, validate and publish eSubmissions based on standardized submission formats comes with its own set of challenges. In addition, managing submissions in multiple regions also requires that all submissions must be compliant with the ICH and regional specifications, which further complicates the overall process.
To ensure compliance, you need an eCTD software solution that enables you to maintain a comprehensive overview of your submission statuses across a number of products within multiple different geographic markets. EXTEDO eCTDmanager is designed to meet exactly these needs and ensure effortless compliance with regulatory requirements.
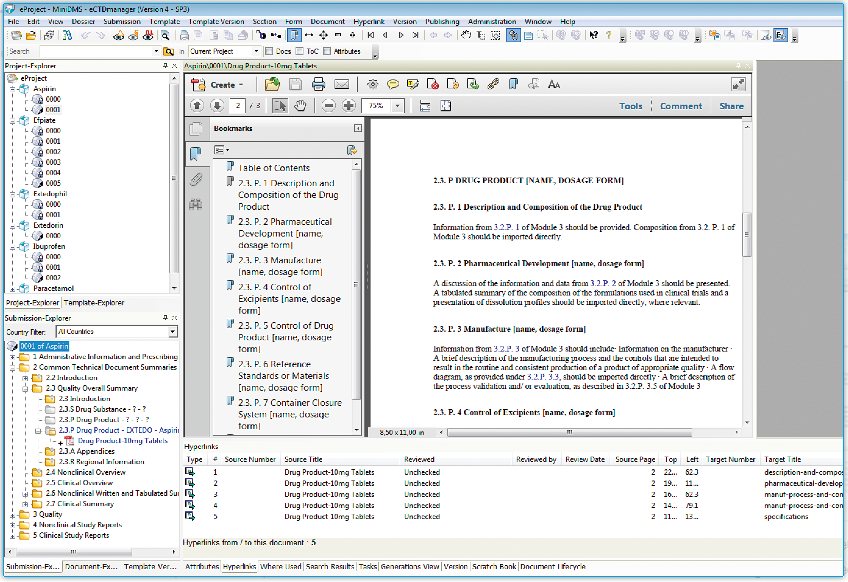
eCTDmanager provides you with a complete regulatory dossier management and assembly solution. Its scalable, all-in-one submission management capabilities meet the requirements for both electronic (like eCTD) and paper submissions. The intuitive user interface is easy to use, and the imported word document will automatically convert to PDF format, allowing you to easily handle electronic submissions.
eCTDmanager Benefits
- Rapid compliance with latest regulatory changes worldwide
- Ability to validate and publish submissions from a single application
- Supports publication of eSubmissions and paper-based submissions in structures and formats for all regions of the world.
- Improves submission quality and consistency.
- Sophisticated management of hyperlinks and bookmarks.
- Conforms to ICH and regional filenames and 21 CFR Part 11
- Minimal hardware requirements, high performance.
- Rapid deployment and system validation.
- Minimum training.

